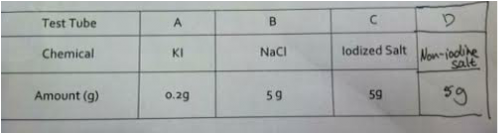
Procedure
1. Label four large test tubes A, B, C and D and add the following reagents to each test tube.
2. Add 5mL of distilled or deionized water to each test tube. Note: the solids in test tubes B, C and D will not fully dissolve. The excess solid is used because KI is present in very small amounts.
3. Add 5 drops of starch solution, followed by 2-3 drops of chlorine water, to each test tube.
4. Compare the color changes observed in each test tube and discuss the qualitative evidence for the presence of iodide in iodized salt.
3. Add 5 drops of starch solution, followed by 2-3 drops of chlorine water, to each test tube.
4. Compare the color changes observed in each test tube and discuss the qualitative evidence for the presence of iodide in iodized salt.
Results
A: Became a light purple color, a translucent solution
B: Some purple in the liquid, salt still white
C: Cloudy, no real visible color change
D: Cloudy, no real visible color change
Test Tubes A and B were the only ones with a real visible color change in this experiment. The reaction shows is there is iodide presence in the substance.
B: Some purple in the liquid, salt still white
C: Cloudy, no real visible color change
D: Cloudy, no real visible color change
Test Tubes A and B were the only ones with a real visible color change in this experiment. The reaction shows is there is iodide presence in the substance.