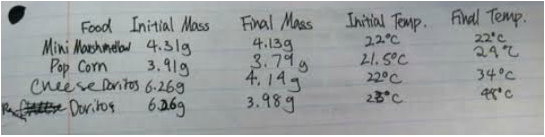Procedure
Alternative Procedure: A "Soda Can" Calorimeter
1. Place a food sample on the food holder. Measure and record the combined mass of the food holder and sample. Place the food holder on a ring stand.
2. Obtain a clean, empty soda can. Measure and record its mass.
3. Add about 50 ml of tap water to the can and measure the combined mass of the can and the water.
4. Bend the top tab on the can up and slide a stirring rod straight through the hole. Suspend the can on a ring stand using a metal ring. Adjust the height of the can so that it is about 2.5 cm above the food holder.
5. Insert a thermometer into the can. Measure and record the initial temperature of the water.
6. Light the food sample and center it under the soda can. Allow the water to be heated until the food sample stops burning. Record the maximum (final) temperature of the water in the can.
7. Measure and record the final mass of the food holder and sample.
8. Clean the bottom of the can and remove any food residue from the food holder. Repeat the procedure with a second food sample.
1. Place a food sample on the food holder. Measure and record the combined mass of the food holder and sample. Place the food holder on a ring stand.
2. Obtain a clean, empty soda can. Measure and record its mass.
3. Add about 50 ml of tap water to the can and measure the combined mass of the can and the water.
4. Bend the top tab on the can up and slide a stirring rod straight through the hole. Suspend the can on a ring stand using a metal ring. Adjust the height of the can so that it is about 2.5 cm above the food holder.
5. Insert a thermometer into the can. Measure and record the initial temperature of the water.
6. Light the food sample and center it under the soda can. Allow the water to be heated until the food sample stops burning. Record the maximum (final) temperature of the water in the can.
7. Measure and record the final mass of the food holder and sample.
8. Clean the bottom of the can and remove any food residue from the food holder. Repeat the procedure with a second food sample.
Data
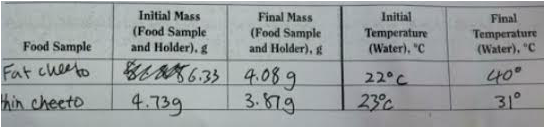
Mass of Empty Soda Can: 13.05g
Mass of Can and Tap Water: 63.08g
Mass of Water: 50.03g
Mass of Can and Tap Water: 63.08g
Mass of Water: 50.03g
- Change in Temperature
- Fat Cheeto - 18 degrees
- Regular Cheeto - 8 degrees
- Marshmallow - no change
- Pop Corn - 2.5 degrees
- Cheese Dorito - 12 degrees
- Cool Ranch Dorito - 25 degrees
- q = m x s x (Change in)T - Calories in Food
- Fat Cheeto:q = 50.03 x 1 x 18 =
- Calories - 900.54
- Kilocalories - .90054
- Nutritional Calories - .90054
- Regular Cheeto: q = 50.03 x 1 x 8
- Calories - 400.24
- Kilocalories - .40024
- Nutritional Calories - .40024
- Marshmallow: q = 50.03 x 1 x 0 (Note: Even though the results are 0, this is simply our interpretation of the results we got. There was no change in temperature in the water because of the burning marshmallow, so if calculated that means no calories.)
- Calories - 0
- Kilocalories - 0
- Nutritional Calories - 0
- Popcorn: q = 50.03 x 1 x 2.5
- Calories - 125.075
- Kilocalories - .125075
- Nutritional Calories - .125075
- Cheese Doritos: q = 50.03 x 1 x 12
- Calories - 600.36
- Kilocalories - .60036
- Nutritional Calories - .60036
- Cool Ranch Dorito: q = 50.03 x 1 x 25
- Calories - 1250.75
- Kilocalories - 1.25075
- Nutritional Calories - 1.25075
- Mass of Food Burned
- Fat Cheeto - 2.25 g
- Regular Cheeto - .86 g
- Marshmallow - .18g
- Popcorn - .12g
- Cheese Doritos - 2.12g
- Cool Ranch Dorito - 2.28g
- Calories per g
- Fat Cheeto - 900.54/2.25 = 400.24/g
- Regular Cheeto - 400.24/.86 = 465.4/g
- Marshmallow - 0/.18 = 0/g
- Popcorn - 125.075/.12 = 1042.29/g
- Cheese Dorito - 600.36/2.12g = 283.19/g
- Cool Ranch Dorito - 1250.75/2.28 = 548.58/g
Results
There was some error in the testing, including no change in temperature as a result of the marshmallow, the snacks falling off of the stands (specifically the Doritos), and the fact that in the Cool Ranch Dorito test, the cork used to hold up the food may have caught on fire for a little bit.